But despite the COVAX deal it may still be at the end of a long queue, given the hundreds of millions of doses of Pfizer mRNA vaccine already pre-purchased by the United States, Japan and the European Union, and similar deals for these and other countries in negotiation with Moderna.Ĭompare this with Germany, where a planned rollout of the Pfizer vaccine to the elderly will start 24 hours after emergency approval, potentially as early as this month. So what should Australia do?Īustralia’s first option will be to buy doses from overseas. 
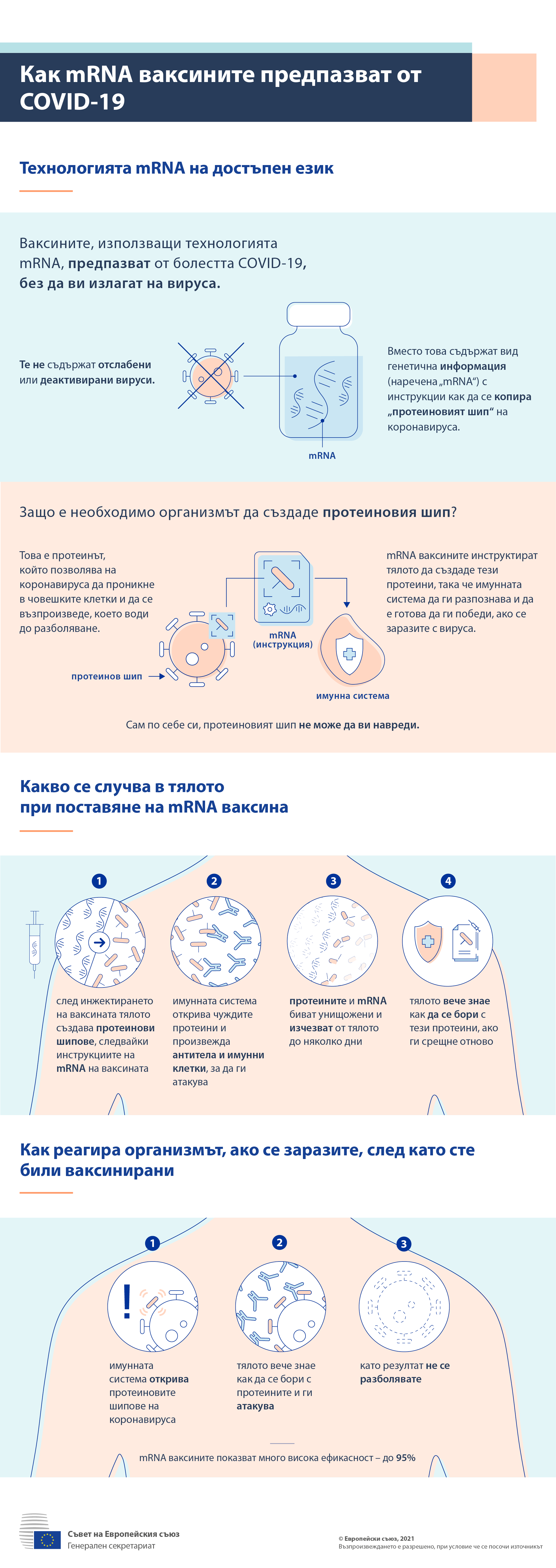
CSL has not declared any desire to establish mRNA manufacturing in Australia at this time. But CSL has issued no public statement about its capacity for Australian production of clinical-grade mRNA vaccines if this, or one of the COVID-19 mRNA candidate vaccines, requires a local supply. The CSL global product pipeline includes an mRNA vaccine against the flu in pre-clinical development. With the federal government prepared to invest A$330 million in research for COVID vaccines and treatment, and mRNA vaccines clearly leading the global race, it’s possible some Australian biotech firms could pivot to mRNA production. It is not publicly known whether any company responded indicating it could make mRNA vaccines. In August, federal science and technology minister Karen Andrews, called on Australian businesses to come forward if they can help with vaccine production and distribution. Concerns raisedĪustralian scientists recently raised concerns about the lack of capacity for mRNA vaccine production. Melbourne-headquartered global biotech firm CSL can make protein-based vaccines, and has expanded its capacity to include DNA/viral vaccines, but not mRNA.ĬSIRO has facilities for making clinical-grade proteins for phase 1 and 2 clinical trials, but not vaccine-grade mRNA, and not at the scale needed for clinical trials, let alone for immunising the entire population. Here's what to expectĪs a COVAX member, Australia will have access to buy and distribute either of these vaccines if successful in clinical trials, and could also license the technology to make the vaccines domestically.īut Australia does not currently have the capacity to manufacture clinical-grade mRNA vaccines. The Moderna and CureVac candidates are both part of the COVAX initiative, a World Health Organisation-sponsored drive to boost vaccine research and give member countries a wider range of potential candidates.Īustralia's just signed up for a shot at 9 COVID-19 vaccines. LUNAR-COV19 (Arcturus/Duke-NUS, Singapore), phase 1-2ĬOVAC1 (Imperial College, UK) and Covidvax (People’s Liberation Army Academy of Military Sciences/Walvax Biotech, China), both in phase 1. MRNA-1273 (Moderna, US) and BNT162 (Pfizer/BioNTech, Germany), both in phase 3 trials There are six mRNA COVID-19 vaccines in clinical trials: Which mRNA vaccines are the frontrunners? So while the production of mRNA is cleaner, it may also be slowed by supply chain issues. This means distribution may be difficult, especially in the developing world.Īnd crucially, most countries - including Australia - don’t have the mRNA manufacturing capability needed to make these vaccines at the required scale. At normal refrigerator temperatures of 2-8℃, they tend to last just a day or two. For example, Moderna’s needs to be kept at -20℃ and Pfizer’s at -70℃. They also need to be stored at very low temperatures. Unlike other vaccines, we do not have years of data on the safety of this type of vaccine to reassure the public. As a new technology, no mRNA vaccine has ever been approved for clinical use. However, mRNA vaccines also have some disadvantages. This means the mRNA can copy itself inside our cells, so protein production lasts longer and, potentially, fewer doses are needed. Some mRNA vaccines, such as Imperial College London’s vaccine now undergoing testing, are even self-replicating. Unlike other types of vaccines made in living cells such as chicken eggs or genetically modified cell cultures, mRNA molecules can be made in an apparatus called a bioreactor. MRNA vaccines can be potentially quicker and cleaner to make than other vaccines. This speed of design is why the COVID-19 mRNA vaccines are current frontrunners, and will probably be the first to get approval by the US Food and Drug Administration. This means mRNA vaccines can be rapidly designed for new viruses or strains. Their production process is almost identical for any possible mRNA. The body then makes these proteins, and the immune system recognises them and mounts an immune response. MRNA vaccines work by delivering instructions to cells to make viral proteins. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
